Microfluidics in Pharmaceutical Nanomanufacturing: Towards Scalable and Controlled Drug Delivery
DOI:
https://doi.org/10.64062/JPGMB.Vol2.Issue2.4Keywords:
- Microfluidics; Nanomanufacturing; Drug delivery; Lipid nanoparticles; Polymeric nanoparticles; Continuous-flow synthesis; Precision nanomedicine.
Abstract
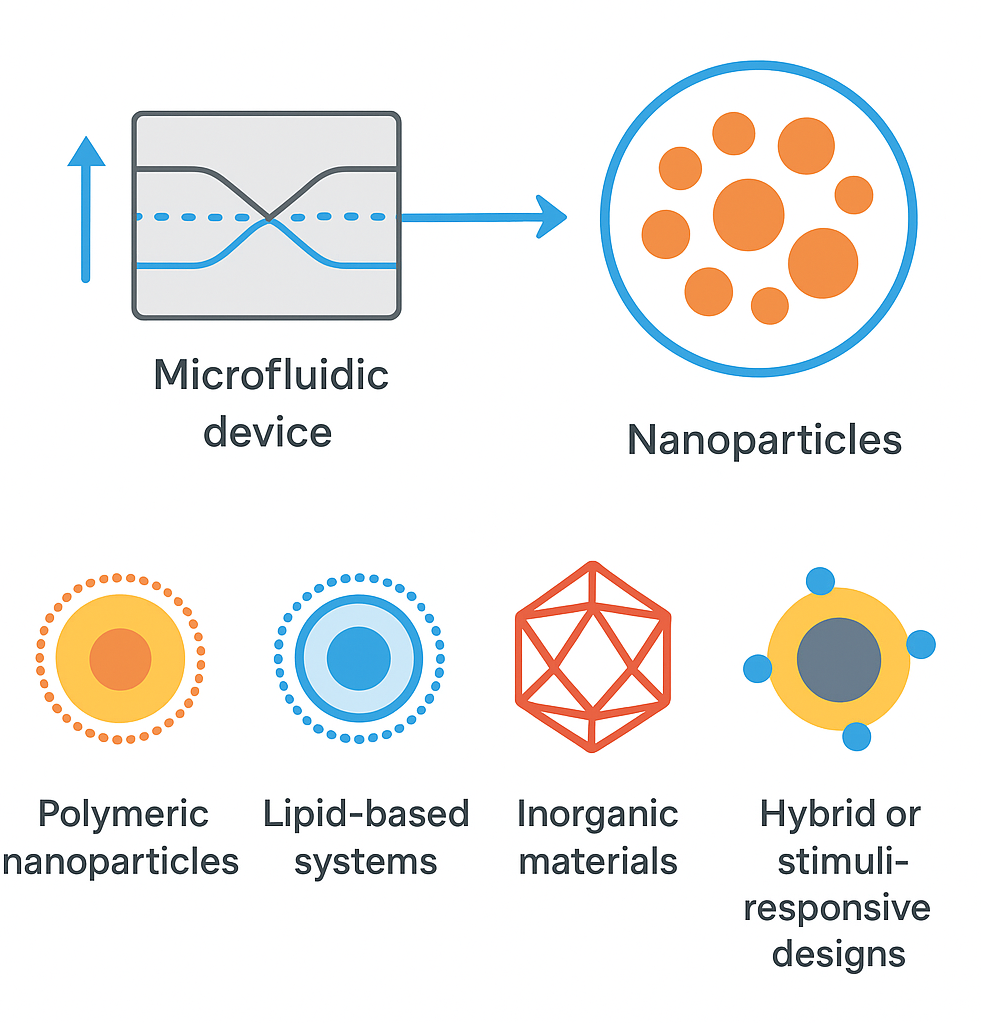
Microfluidic technology has emerged as a transformative platform in pharmaceutical nanomanufacturing, offering unprecedented control over the synthesis of nanoscale drug delivery systems. Traditional bulk manufacturing techniques often suffer from poor reproducibility, wide size distributions, and limited scalability, restricting their utility for clinical-grade nanoparticle production. In contrast, microfluidics enables precise manipulation of fluids under laminar flow, facilitating diffusion-driven mixing, rapid nucleation, and consistent particle formation across diverse nanocarriers including polymeric nanoparticles, lipid-based systems, inorganic materials, and hybrid or stimuli-responsive designs. The integration of real-time analytics, automated process optimization, and advanced microreactor geometries further enhances the ability to tailor nanoparticle properties such as size, polydispersity, and surface functionality. Recent advances in flow rate control, parallelization, and device engineering have accelerated the transition of microfluidics from laboratory research to scalable industrial production, exemplified by its successful application in large-scale mRNA lipid nanoparticle manufacturing. Emerging synergies with AI/ML tools, organ-on-chip platforms, 3D printing, and IoT-enabled continuous-flow systems are pushing microfluidic nanomanufacturing toward fully autonomous, high-throughput factories. Despite challenges related to device fouling, material compatibility, cost, and regulatory frameworks, microfluidics is poised to become a cornerstone of next-generation drug delivery, enabling highly controlled, customizable, and clinically translatable nanomedicine.
References